DFT simulations and Raman spectroscopy properties of Capsaicin
DOI:
https://doi.org/10.31349/RevMexFis.72.030503Keywords:
Capsaicin; DFT calculations; Raman spectroscopy; conformational analysisAbstract
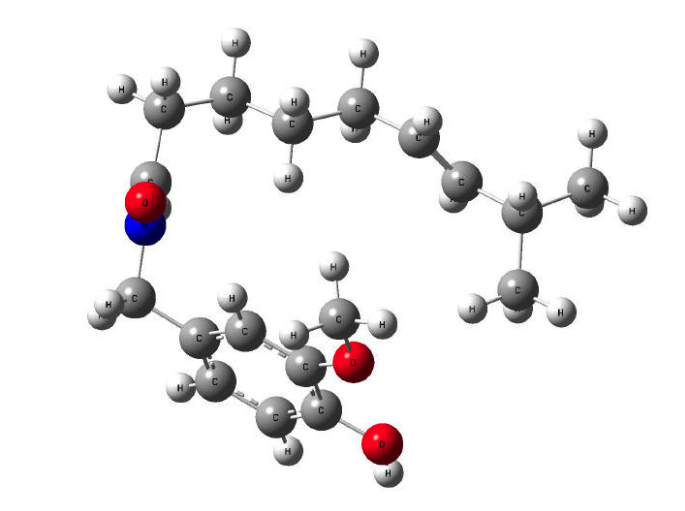
A theoretical study of capsaicin was conducted using density functional theory (DFT) with the B3LYP and CAM-B3LYP functionals, combined with the 6-31G(d), 6-311+G(d,p), LanL2DZ, and CC-pVDZ basis sets. The study analyzed theoretical vibrational modes, Raman spectra, conformational energies, and global minima in geometric optimization. Results indicate that the LanL2DZ basis set provides the lowest conformational energies and the fastest computation times, whereas 6-31G(d) yields higher EPM values in VEDA. The CAM-B3LYP functional, particularly when combined with advanced basis sets such as 6-311+G(d,p) and CC-pVDZ, yields more accurate electronic property predictions. Overall, CAM-B3LYP/6-311+G(d,p) offers the best compromise between computational efficiency and spectral accuracy, while B3LYP/CC-pVDZ remains suitable for preliminary analyses.
Downloads
References
E. K. Nelson and E. Dawson, The constitution of capsaicin, the pungent principle of capsicum. J. Am. Chem. Soc. 45 (1923) 2179, https://doi.org/10.1021/ja01662a023 DOI: https://doi.org/10.1021/ja01662a023
E. Späth and S.F. Darling, Synthese des Capsaicins, Ber. dtsch. Chem. Ges. A/B 63(1930) 737, https://doi.org/10.1002/cber.19300630331 DOI: https://doi.org/10.1002/cber.19300630331
National Center for Biotechnology Information. PubChem Compound Summary for CID 1548943, Capsaicin. PubChem, https://pubchem.ncbi.nlm.nih.gov/compound/Capsaicin
C. Kim et al., Capsaicin exhibits anti-inflammatory property by inhibiting IkB-a degradation in LPS-stimulated peritoneal macrophages, Cellular Signalling 15 (2003) 299, https://doi.org/10.1016/S0898-6568(02)00086-4 DOI: https://doi.org/10.1016/S0898-6568(02)00086-4
M. H. Yang, S. H. Jung, G. Sethi, and K. S. Ahn, Pleiotropic Pharmacological Actions of Capsazepine, a Synthetic Analogue of Capsaicin, against Various Cancers and Inflammatory Diseases, Molecules, 24 (2019) 995, https://doi.org/10.3390/molecules24050995 DOI: https://doi.org/10.3390/molecules24050995
S. Liu et al., Label-free SERS strategy for rapid detection of capsaicin for identification of waste oils, Talanta 245 (2022) 123488, https://doi.org/10.1016/j.talanta.2022.123488 DOI: https://doi.org/10.1016/j.talanta.2022.123488
K. Tian et al., Rapid identification of gutter oil by detecting the capsaicin using surface enhanced Raman spectroscopy. J. Raman Spectrosc. 49 (2018) 472, https://doi.org/10.1002/jrs.5306 DOI: https://doi.org/10.1002/jrs.5306
Z. Liu, S. Yu, S. Xu, B. Zhao, and W. Xu, Ultrasensitive detection of capsaicin in oil for fast identification of illegal cooking oil by SERRS. Acs Omega 2 (2017) 8401, https://doi.org/10.1021/acsomega.7b01457 DOI: https://doi.org/10.1021/acsomega.7b01457
A. Ekhlas, N. A. H. Zahra, and A. A. Jenan, FT-IR Identification of Capsaicin from callus and seedling of chilli pepper plants Capsicum annuum L. in vitro, Int. J. Multidiscip. Curr. Res. 4 (2016) 1144, https://ijmcr.com/index.php/ijmcr/article/view/04.06.09/672
M. C¸ ınar, B. Alim, Z. Alim, E. S¸akar, Determination of the molecular structure and spectroscopic properties of capsaicin. Rad. Phys. Chem. 208 (2023) 110879, http://dx.doi.org/10.2139/ssrn.4292963 DOI: https://doi.org/10.2139/ssrn.4292963
G. P. S. Mol, D. Aruldhas, I. H. Joe, S. Selvaraj, and A. G. Nadh, Modeling the structural and reactivity properties of capsaicin [(E)-N-[(4-hydroxy-3-methoxyphenyl)methyl]- 8-methylnon-6-enamide] wavefunction-dependent properties, pharmacokinetics, in-silico analysis, and molecular dynamics simulation, J. Mol. Struct. 1304 (2024) 137591, https://doi.org/10.1016/j.molstruc.2024.137591 DOI: https://doi.org/10.1016/j.molstruc.2024.137591
E. Romano et al., Identification of cholesterol in different media by using the FT-IR, FT-Raman and UV-visible spectra combined with DFT calculations, J. Mol. Liq. 403 (2024) 124879, https://doi.org/10.1016/j.molliq.2024.124879 DOI: https://doi.org/10.1016/j.molliq.2024.124879
M. Karabacak, M. Cinar, and M. Kurt, DFT based computational study on the molecular conformation, NMR chemical shifts and vibrational transitions for N-(2-methylphenyl) methanesulfonamide and N-(3-methylphenyl) methanesulfonamide, J. Mol. Struct. 968 (2010) 108, https://doi.org/10.1016/j.molstruc.2010.01.033 DOI: https://doi.org/10.1016/j.molstruc.2010.01.033
D. Zhang et al., SERS determination of hydroxy-α-sanshool in spicy hotpot seasoning: The strategy to restrain the interference of capsaicin and its mechanism, Food Chem. 413 (2023) 135644, https://doi.org/10.1016/j.foodchem.2023.135644 DOI: https://doi.org/10.1016/j.foodchem.2023.135644
P. Siudem, K. Paradowska, J. Bukowicki, Conformational analysis of capsaicin using 13C, 15N MAS NMR, GIAO DFT and GA calculations, J. Mol. Struct. 1146 (2017) 773, https://doi.org/10.1016/j.molstruc.2017.05.142 DOI: https://doi.org/10.1016/j.molstruc.2017.05.142
M. T. Bilkan, Ş. Yurdakul, Z. Demircioǧlu, O. Büyükgüngör, Crystal structure, FT-IR, FT-Raman and DFT studies on a novel compound [C10H9N3]4AgNO3, J. Organomet. Chem. 805 (2016) 108, https://doi.org/10.1016/j.jorganchem.2016.01.014 DOI: https://doi.org/10.1016/j.jorganchem.2016.01.014
J. S. P. P. Leela, R. Hemamalini, S. Muthu and A. A. AlSaadi, Spectroscopic investigation (FTIR spectrum), NBO, HOMO-LUMO energies, NLO and thermodynamic properties of 8-Methyl-N-vanillyl-6-nonenamideby DFT methods, Spectrochim. Acta A 146 (2015) 177, https://doi.org/10.1016/j.saa.2015.03.027 DOI: https://doi.org/10.1016/j.saa.2015.03.027
M. D. L. Reyes-Escogido, E. G. Gonzalez-Mondragon, and E. Vazquez-Tzompantzi, Chemical and pharmacological aspects of Capsaicin, Molecules 16 (2011) 1253, https://doi.org/10.3390/molecules16021253 DOI: https://doi.org/10.3390/molecules16021253
N. D. Kambaine, D. M. Shadrack, and S. A.H. Vuai, Conformations and stability of Capsaicin in bulk solvents: A molecular dynamics study, J. Mol. Liq. 345 (2022) 117794, https://doi.org/10.1016/j.molliq.2021.117794 DOI: https://doi.org/10.1016/j.molliq.2021.117794
M.J. Frisch et al., Gaussian 09, (2009)
A. D. Becke, Density-functional thermochemistry. III. The role of exact exchange, J. Chem. Phys. 98 (1993) 5648, https://doi.org/10.1063/1.464913 DOI: https://doi.org/10.1063/1.464913
C. Lee, W. Yang and R. G. Parr, Development of the ColleSalvetti correlation-energy formula into a functional of the electron density, Phys. Rev. B 98 (1988) 785, https://doi.org/10.1103/PhysRevB.37.785 DOI: https://doi.org/10.1103/PhysRevB.37.785
X. Wu, S. Gao, J. S. Wang, H. Wang, Y. W. Huanga and Y. Zhaod, The surface-enhanced Raman spectra of aflatoxins: spectral analysis, density functional theory calculation, detection and differentiation, Analyst 137 (2012) 4226, https://doi.org/10.1039/C2AN35378D DOI: https://doi.org/10.1039/c2an35378d
T. Yanai, D. P. Tew, and N. C. Handy, A new hybrid exchangecorrelation functional using the Coulomb-attenuating method (CAM-B3LYP). Chem. Phys. Lett. 393 (2004) 51, https://doi.org/10.1016/j.cplett.2004.06.011 DOI: https://doi.org/10.1016/j.cplett.2004.06.011
T. H. Dunning, Gaussian basis sets for use in correlated molecular calculations. I. The atoms boron through neon and hydrogen. J. Chem. Phys. 90 (1989) 1007, https://doi.org/10.1063/1.456153 DOI: https://doi.org/10.1063/1.456153
P. J. Hay and W. R. Wadt, Ab initio effective core potentials for molecular calculations. Potentials for the transition metal atoms Sc to Hg. J. Chem. Phys. 82 (1985) 270, https://doi.org/10.1063/1.448547 DOI: https://doi.org/10.1063/1.448799
K. A. Peterson and T. H. Dunning, Accuracy of correlation consistent basis sets for transition metal and first-row atoms. J. Chem. Phys. 117 (2002) 10548, https://doi.org/10.1063/1.1522883 DOI: https://doi.org/10.1063/1.1520138
M. H. Jamroz, Vibrational energy eistribution analysis ´ (VEDA): Scopes and limitations, Spectrochim. Acta A 114 (2013) 220, https://doi.org/10.1016/j.saa.2013.05.096 DOI: https://doi.org/10.1016/j.saa.2013.05.096
M. Meenu, E. A. Decker, and B. Xu, Application of vibrational spectroscopic techniques for determination of thermal degradation of frying oils and fats: a review. Crit. Rev. Food Sci. Nutr. 62 (2021) 5744, https://doi.org/10.1080/10408398.2021.1891520 DOI: https://doi.org/10.1080/10408398.2021.1891520
J. A. Antunes et al., Study on optical, electrochemical and thermal properties of the Meldrum acid 5-aminomethylene derivative, Vib. Spectrosc. 112 (2021) 103188, https://doi.org/10.1016/j.vibspec.2020.103188 DOI: https://doi.org/10.1016/j.vibspec.2020.103188
R. M. Silverstein, G. C. Bassler and T. C. Morril Spectrometric Identification of Organic Compounds John Wiley, New York (1991)
C. A. Aguirre-Téllez, Q. S. Martins, and J. J. B. Ortega, Breve introducción al estudio Raman de partículas cargadas. Rev. Ingenio 20 (2023) 1, https://doi.org/10.22463/2011642X.3308 DOI: https://doi.org/10.22463/2011642X.3308
M. J. Caterina, M. A. Schumacher, M. Tominaga, T. A. Rosen, J. D. Levine and D. Julius, The Capsaicin receptor: a heatactivated ion channel in the pain pathway. Nature 389 (1997) 816, https://doi.org/10.1038/39807 DOI: https://doi.org/10.1038/39807
D. H. Kwon, F. Zhang, Y. Suo, J. Bouvette, M. J. Borgnia, and S. Lee, Heat-dependent opening of TRPV1 in the presence of capsaicin. Nat. Struct. Mol. Biol. 28 (2021) 554, https://doi.org/10.1038/s41594-021-00616-3 DOI: https://doi.org/10.1038/s41594-021-00616-3
A. Abkari, I. Chaabane, and K. Guidara, DFT (B3LYP/LanL2DZ and B3LYP/6311G+(d,p)) comparative vibrational spectroscopic analysis of organic–inorganic compound bis(4-acetylanilinium) tetrachlorocuprate(II), Physica E 81 (2016) 136, https://doi.org/10.1016/j.physe.2016.03.010 DOI: https://doi.org/10.1016/j.physe.2016.03.010
V.K. Shen, D.W. Siderius, W.P. Krekelberg, and H.W. Hatch, Eds., NIST Standard Reference Simulation Website, NIST Standard Reference Database, National Institute of Standards and Technology, Gaithersburg MD, 20899, https://cccbdb.nist.gov/hartreex.asp
A. Alberti, V. Galasso, B. Kovač, A. Modelli, and F. Pichierri, Probing the Molecular and Electronic Structure of Capsaicin: A Spectroscopic and Quantum Mechanical Study, J. Phys. Chem. 112 (2008) 5700, https://doi.org/10.1021/jp801890g DOI: https://doi.org/10.1021/jp801890g
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Q. S. Martins, D. L. L. Oliveira

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Authors retain copyright and grant the Revista Mexicana de Física right of first publication with the work simultaneously licensed under a CC BY-NC-ND 4.0 that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.





